Atoms are composed of protons, neutrons and electrons. The protons and neutrons occupy the nucleus of an atom and give it its mass. The number of protons identifies the type of atom that it is - referred to as an element. Electrons are located outside of the nucleus and have the opposite type of charge as the protons. If the numbers of protons and electrons are equal, then the atom is neutral.
Getting your Trinity Audio player ready...
Hold down the T key for 3 seconds to activate the audio accessibility mode, at which point you can click the K key to pause and resume audio. Useful for the Check Your Understanding and See Answers.
Subatomic Particles - helpApprentice
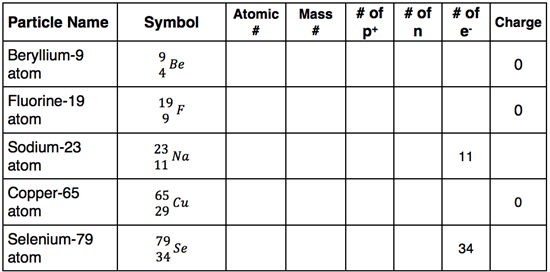
The Apprentice Difficulty Level involves the completion of a table. There are four different versions of the table. Here is one of the versions.
Version 1:
Complete the following table.