Not all solutions are created equal. Some are more concentrated than others. Molarity is one means of expressing how concentrated a solution is with solute. Molarity is defined as the number of moles of solute per liter of solution. Its units are mole/liter, sometimes abbreviated with a capital M. Knowledge of the moles of solute and the liters of solution allows one to calculate molarity by division.
Getting your Trinity Audio player ready...
Hold down the T key for 3 seconds to activate the audio accessibility mode, at which point you can click the K key to pause and resume audio. Useful for the Check Your Understanding and See Answers.
Molarity Calculations - help5
There are three very similar versions of questions in this Question Group. One of the versions is shown below.
Version 1 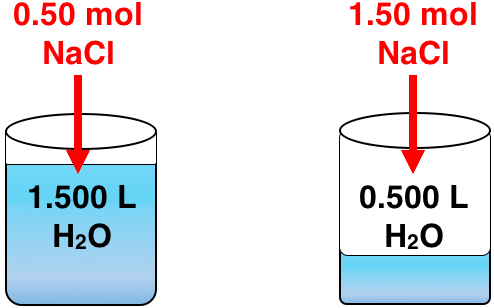
The solute concentration in Solution _______ is greater than the solute concentration in Solution _______ by a factor of ____________.
Version 1
Tap on the fields to accurately complete the statement regarding Solutions A and B.

The solute concentration in Solution _______ is greater than the solute concentration in Solution _______ by a factor of ____________.